Our mission is to increase access to x-ray crystallography and the determination of 3D atomic structures of biological macromolecules. The purpose is to grow the number of structural studies conducted and funded and to increase the results and deliverables generated by this methodology within Minnesota.
Why Determine Structures??
Biological molecules can be very large and comlicated. The structure of these large macromolecules forms the framework for understanding the function. Without the structure, we are like the 6 blind men examining and elephant, each discovering a piece here and there, but not being able to get the complete picture.

Crystallography is Used to Determine the Structures of the Largest Macromolecules
The light microscope makes images using lenses to collect and focus visible light scattered off a sample. Visible light has wavelengths of about 10-5 cm. While this is very small, it is still thousands of times too large to image macromolecules which are measured in Angstroms (1 Angstrom = 10-8 cm). Since x-rays have wavelengths around an Angstrom, they can be used to “see” macromolecules. Crystallography is the technique of determining structures from the x-ray diffraction patterns generated by crystals of a substance. In some ways it is like using a microsope to view an object.
No single technique produces more detailed data about a macromolecule than crystallography. The resulting atomic structures allow assembly of separate observations into a coherent whole and thus are crucial for understanding the biochemical and biophysical roles of a macromolecule in their interactions with other molecules, in cellular function and in disease. Another driving force is the rational design of therapeutically important inhibitors and activators, drugs, based on deciphering high-resolution conformations of lead compounds and the molecules to which they bind. One need only look back at major discoveries in the last century, e.g., Watson & Crick and DNA, Perutz and hemoglobin, Huber and photosynthetic reaction center, MacKinnon and ion channels, and Kornberg and RNA polymerase, to see the significance of crystallography.
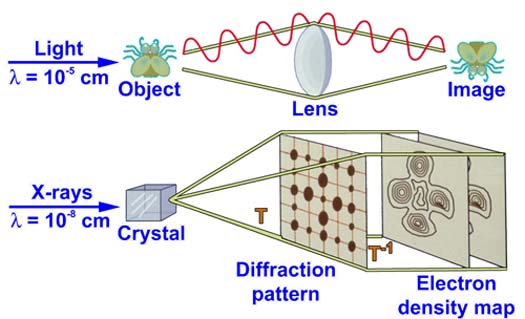
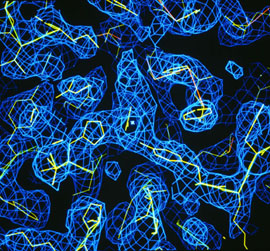
1. The x-rays interact with the electrons in the material and are scatteted and collected in a way that allows the scientist to use mathematics and computers to reconstruct a map of the electron density of the substance.
2. The electron map (shown in blue on the right) allows the scientist to determine where most of the atoms (shown in yellow) of the macromolecule are located.
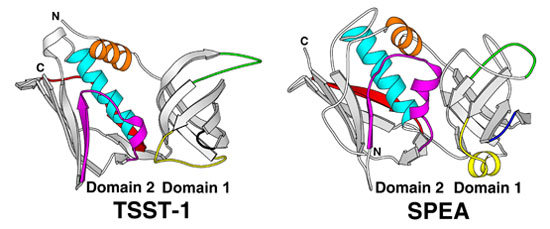
3. Using and computer and knowing the bond lengths for the atoms a scientis can build a model of the marcomolecule. The models below are for Toxic Shock Syndrom Toxin 1 (TSST-1) and Scalet Fever Toxin (Spea). Both of these structres were determined here in Minnesota. Work on deleveloping a vaccine is in progress.
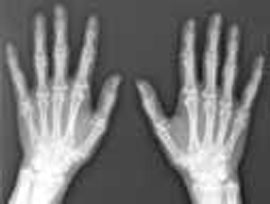
4. X-rays only interact weakly with proteins, only our bones are visible with any detail in the image on the left, not the muscles and other tissues. To "see a macromolecule" we need to amplify the signal by arranging molecules in a regular pattern which acts to reinforces the scattering
5. The use of crystals of a protein in which the molecule are ordered gives enough amplification of the signal to allow the structure to be determined. In a protein crystal the mole
cules are arranged in an orderly repeating pattern extending in all three spatial dimensions.
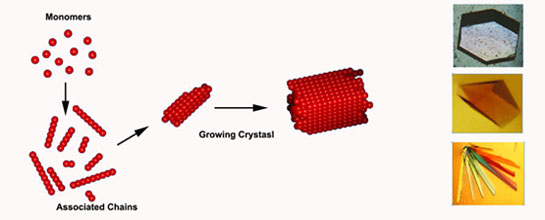
6. Protein crystals do not occur naturally and must be grown by hand. Protein crystals are grow by trail and error sampling of hundreds or even thousands of different conditions. The use of a robot makes this task simpler, more cost effective, and reliable.

A Famous Example
- DNA was believed to be chemically too simple to contain the amount of information need to direct development
- Proteins which were complex were believed to be the substance composing genes
- Watson and Crick proposed the now famous double helix DNA model which suggested how the DNA molecule could code for an enormous amount of information
